|
Nevertheless, before the mass spectrometry is applied, the sample is digested by trypsin into short peptides and then fractionated by isoelectric focusing into so called fractions which allows to reduce MS analysis complexity. Therefore, 2D-PAGE has been today replaced in many cases by gel-free techniques such as high-throughput mass spectrometry (MS). 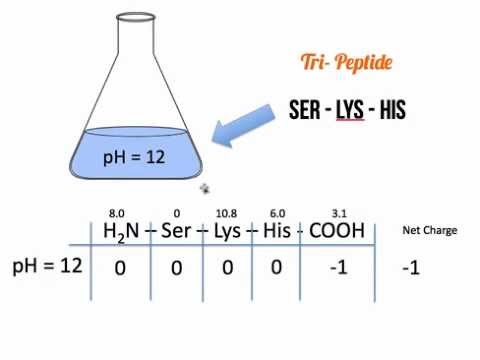
Unfortunately, 2D-PAGE suffers from several intrinsic technical problems (e.g., performs poorly for very large, very small, extremely acidic or basic proteins). One of the oldest, but still widely used technique is 2-D polyacrylamide gel electrophoresis (2D-PAGE), where proteins are separated in two dimensions on a gel and identified using estimated molecular weight and isoelectric point ( pI is the pH value at which the net charge of a macromolecule is zero, and therefore its electrophoretic mobility is stopped). Over the years, many techniques were introduced to allow to accomplish the task. 
The procedure relies on physicochemical properties of amino acids such as a molecular mass or a charge. Next, individual proteins are separated and finally identified. This article was reviewed by Frank Eisenhaber and Zoltán GáspáriĪnalysis of proteins starts from the heterogeneous mixture (lysate) from which protein fraction needs to be isolated. The IPC service is freely available at Peptide and protein datasets used in the study and the precalculated pI for the PDB and some of the most frequently used proteomes are available for large-scale analysis and future development. Moreover, the prediction of pI using the IPC pKa’s leads to fewer outliers, i.e., predictions affected by errors greater than a given threshold. According to the presented benchmarks, the newly developed IPC pKa sets outperform previous algorithms by at least 14.9 % for proteins and 0.9 % for peptides (on average, 22.1 % and 59.6 %, respectively), which corresponds to an average error of the pI estimation equal to 0.87 and 0.25 pH units for proteins and peptides, respectively. Here, I present the Isoelectric Point Calculator (IPC), a web service and a standalone program for the accurate estimation of protein and peptide pI using different sets of dissociation constant ( pKa) values, including two new computationally optimized pKa sets. Additionally, pI estimation can be helpful during protein crystallization trials. It is extremely important to understand the physical forces behind a peptide bond, as this allows scientists to design accurate, predictive models of three-dimensional protein structures.Accurate estimation of the isoelectric point ( pI) based on the amino acid sequence is useful for many analytical biochemistry and proteomics techniques such as 2-D polyacrylamide gel electrophoresis, or capillary isoelectric focusing used in combination with high-throughput mass spectrometry. A mistake in the translation process can lead to protein mis-folding, and in turn, disease. Proteins can be as small as forty-four amino acids, or as large as thirty-five thousand. Peptide bonds are made within ribosomes during a process called «translation» to form polypeptides, which then undergo various molecular processing and modification, before folding into a three-dimensional shape, which we call a protein. For instance, there is currently much interest in antibody-drug conjugates these pair fragmented antibodies with pharmacologically active compounds in order to specifically target cancer tumors, among other things. The ability to predictably split peptide bonds is vital to a number of different fields of study. Reversing a peptide bond without an enzyme is extremely difficult, thus this process is usually mediated by an enzyme called a protease, such as subtilisin, which is frequently added to laundry detergent to cleave many protein contaminants. It may therefore be counterintuitive to learn that peptide bonds are quite stable kinetically: the lifetime of a peptide bond in aqueous solution is approximately 1000 years. Hence, the biosynthesis of a peptide bond requires an input of free energy. One interesting thing to note is that the equilibrium of this reaction lies on the side of hydrolysis rather than synthesis. For most peptides the cis-form is about 1000 times less stable than the trans-form. In such cases, the cis form is more stable than usual since the proline side-chain offers less of a hindrance. 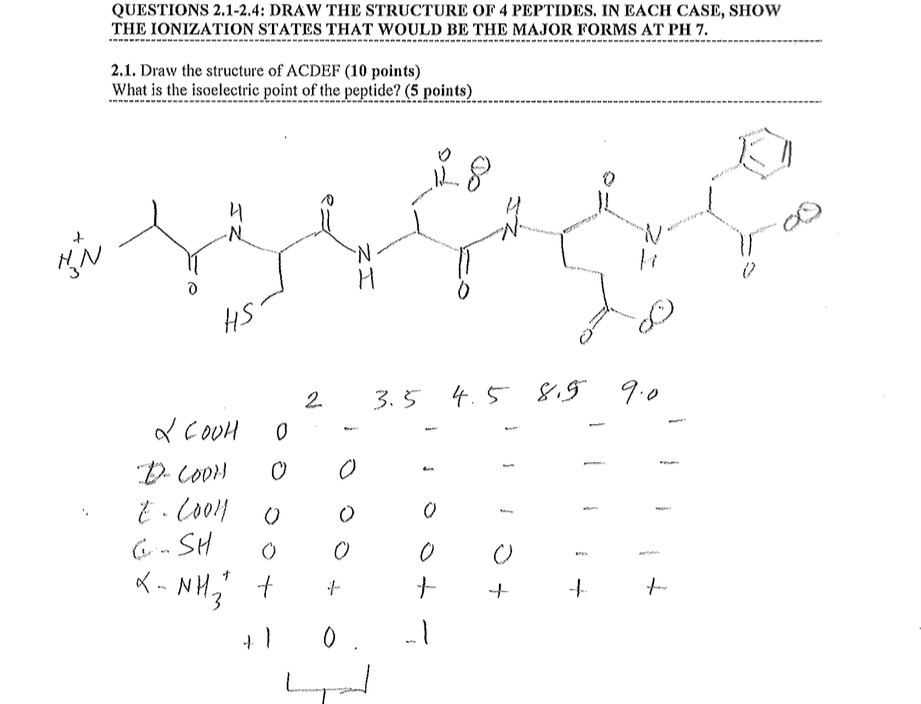
However, cis forms can occur in peptide bonds that precede a proline residue. In naturally occurring peptides most peptide bonds are in the trans configuration.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
